|
5/30/2023 0 Comments Red bull caffeine content NoDoz) must include warnings,” write the Johns Hopkins researchers, “whereas a 500 mg energy drink can be marketed with no such warnings and no information on caffeine dose amount in the product.”Īs early as 2009, sports and medical organizations began issuing position statements discouraging energy drink consumption by young people. “It is a striking inconsistency that, in the US an stimulant medication containing 100 mg of caffeine per tablet (e.g. Drugs with caffeine require warning labels, but dietary supplements don’t. When energy drinks first appeared on the American market in the late 1990s and early 2000s, some manufacturers claimed the products were neither drugs nor conventional foods, but dietary supplements.  
“If caffeine had not been accepted as a flavor enhancer, but had been regarded as a psychoactive ingredient,” write the Johns Hopkins researchers, “soft drinks might have been regulated by the FDA as drugs”-which are subject to additional regulations. 02%, or roughly 71 milligrams per 12-ounce serving. The FDA approved caffeine, but limited the maximum content of cola-type soft drinks to. The manufacturers, however, claimed the caffeine was a flavor enhancer. In 1980, citing health concerns, the FDA proposed to eliminate caffeine from soft drinks, which are regulated as foods. Researchers from the Johns Hopkins School of Medicine, in a review published in the journal Drug and Alcohol Dependence, note that lack of consistency is partly due to our long love affair with drinks in which caffeine is naturally occurring, including coffee and tea. 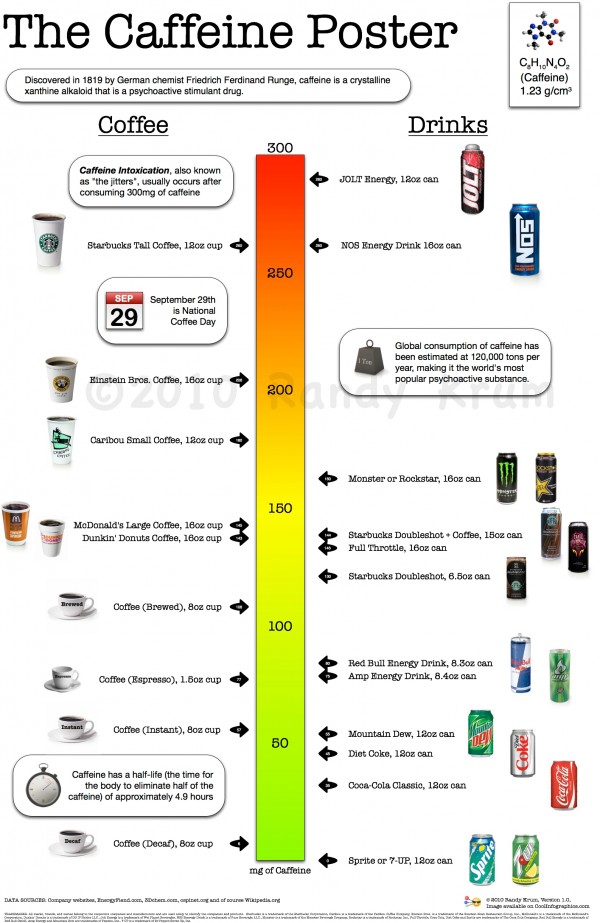
All three major energy drink makers now have most of their products regulated as foods, rather than dietary supplements-though that wasn’t always the case. Though it offers some guidance, the FDA allows manufacturers of liquid products to decide on their own whether to market their products as dietary supplements, or as conventional foods and beverages, which carry differing regulatory requirements. Historically, government agencies such as the US Food and Drug Administration have struggled to regulate beverages with added caffeine. The reasons are a mix of lax regulation, the use of caffeine as a sports performance enhancer among adults, and a bit of scientific uncertainty.Īccording to sports cardiologist John Higgins, a professor at McGovern Medical School at UTHealth in Houston, there is also another factor: “very, very intelligent advertising.” “Age-gating is an incredibly powerful tool,” Luppino said, and should be reserved for “inherently dangerous products” like nicotine.Īs the regulatory status of energy drinks continues to be debated, a growing number of consumers and public health advocates are asking why and how a product loaded with caffeine and other stimulants became so popular among young people. At the Connecticut hearing, the head of public affairs for Red Bull North America, Joseph Luppino, maintained that there is no scientific justification to regulate energy drinks differently than other caffeine-containing beverages such as soda, coffee, and tea-particularly when some coffeehouses serve coffee with a caffeine content exceeding that of a can of Red Bull. Of these, approximately 1,500 were children aged 12 to 17, although the number of visits from this age group increased only slightly over the four years.įor their part, energy drink manufacturers argue that they are being unfairly targeted. A US government report found that from 2007 to 2011, the number of emergency department visits involving energy drinks more than doubled, to nearly 21,000. 
These recommendations are based on concerns about health problems that, although rare, can occur after consumption, including seizures, delirium, rapid heart rate, stroke, and even sudden death. This popularity is in marked contrast to the recommendations of groups like the American Academy of Pediatrics and the American College of Sports Medicine, who say youth should forgo these products entirely. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
